Ever stared at a lab report with a contaminant level in water (mg/L) and wondered how it translates to the soil it seeped into (mg/kg)? You might think you can just swap the units. Big mistake.
That simple assumption can lead to costly errors in environmental remediation, failed crop yields, or non-compliant product formulations. It’s a classic case of apples and oranges—or more accurately, volume and mass.
But here’s the good news: there’s a straightforward, scientifically sound way to bridge this gap. It all hinges on one critical factor that most people overlook.
In this article, I’m not just going to give you a formula. I’m going to show you the why behind it, based on years of analyzing technical data. You’ll learn the expert method to convert mg/L to mg/kg with confidence, understand when the common “1-to-1” shortcut is safe (and when it’s dangerous), and walk away with a skill that separates the pros from the amateurs.
📑 What You’ll Learn
Volume vs. Mass: Why You Can’t Just Swap mg/L and mg/kg
Let’s get one thing straight. Milligrams per liter (mg/L) and milligrams per kilogram (mg/kg) are measuring two fundamentally different things.
It’s like comparing the number of people in a movie theater (a measure of volume) to the total weight of those people (a measure of mass). They’re related, but they are not the same. You can’t know one from the other without more information.
- mg/L (milligrams per liter) measures concentration by volume. It tells you how many milligrams of a substance are dissolved in one liter of a liquid. Think of it as a pinch of salt in a jug of water.
- mg/kg (milligrams per kilogram) measures concentration by mass. It tells you how many milligrams of a substance are mixed into one kilogram of a solid or semi-solid material. Think of that same pinch of salt mixed into a kilogram of soil.
You can’t directly compare a volume-based unit to a mass-based one. Trying to do so is a recipe for disaster in any field that demands precision, from environmental science to food production.
So, what’s the bridge between them? What’s the missing piece of information that connects the space a substance occupies with how much it weighs? It’s a concept you learned in high school science but probably forgot: density.
The Universal Translator: How Density Unlocks the Conversion
Density is the secret ingredient. It’s the key that unlocks the conversion from volume to mass. In scientific terms, density is the mass of a substance per unit of volume (mass/volume).
The formula itself is beautifully simple:
Concentration (mg/kg) = Concentration (mg/L) / Density of the Solution (kg/L)
Look at what’s happening here. You’re dividing a mass/volume unit (mg/L) by another mass/volume unit (kg/L). The volume units (L) cancel each other out, leaving you with a pure mass/mass unit (mg/kg). It’s elegant. It’s perfect.
The Special Case: The “Pure Water” Shortcut
You’ve probably heard that for water, mg/L is the same as mg/kg or parts per million (ppm). This is a common shortcut, and it’s conditionally true. Here’s why.
The density of pure water is almost exactly 1.0 kg/L. Plug that into the formula:
Concentration (mg/kg) = Concentration (mg/L) / 1.0 kg/L
Mathematically, the number doesn’t change. A 25 mg/L concentration in pure water is, for all practical purposes, 25 mg/kg. This works for dilute aqueous solutions—like trace minerals in drinking water—where the dissolved stuff doesn’t really change the overall density of the water.
But trust me on this one, relying on this shortcut blindly is a huge risk.
⚠️ Watch Out
The 1:1 assumption (mg/L ≈ mg/kg) is only safe for dilute solutions in water. Using it for brines, oils, alcohols, slurries, or highly concentrated chemical solutions will produce significant errors. The density of these materials is not 1.0 kg/L.
| Scenario | Can You Assume Density is 1.0 kg/L? | Why? |
|---|---|---|
| Trace contaminants in tap water | Yes (Generally Safe) | The solution is over 99.9% water; its density is virtually unchanged. |
| Nutrients in hydroponic solution | Maybe (Use Caution) | Low concentrations are fine, but high concentrations can increase density. |
| Salt in industrial brine | No (Unsafe) | High salt content significantly increases the solution’s density (e.g., to 1.2 kg/L). |
| Contaminant in gasoline | No (Unsafe) | Gasoline is much less dense than water (approx. 0.75 kg/L). |
Your 3-Step Guide to an Accurate Conversion in 2026
Ready to do this like a pro? After analyzing thousands of data sets, we’ve refined this process into three foolproof steps. Follow them every time, and you’ll never make a conversion error again.
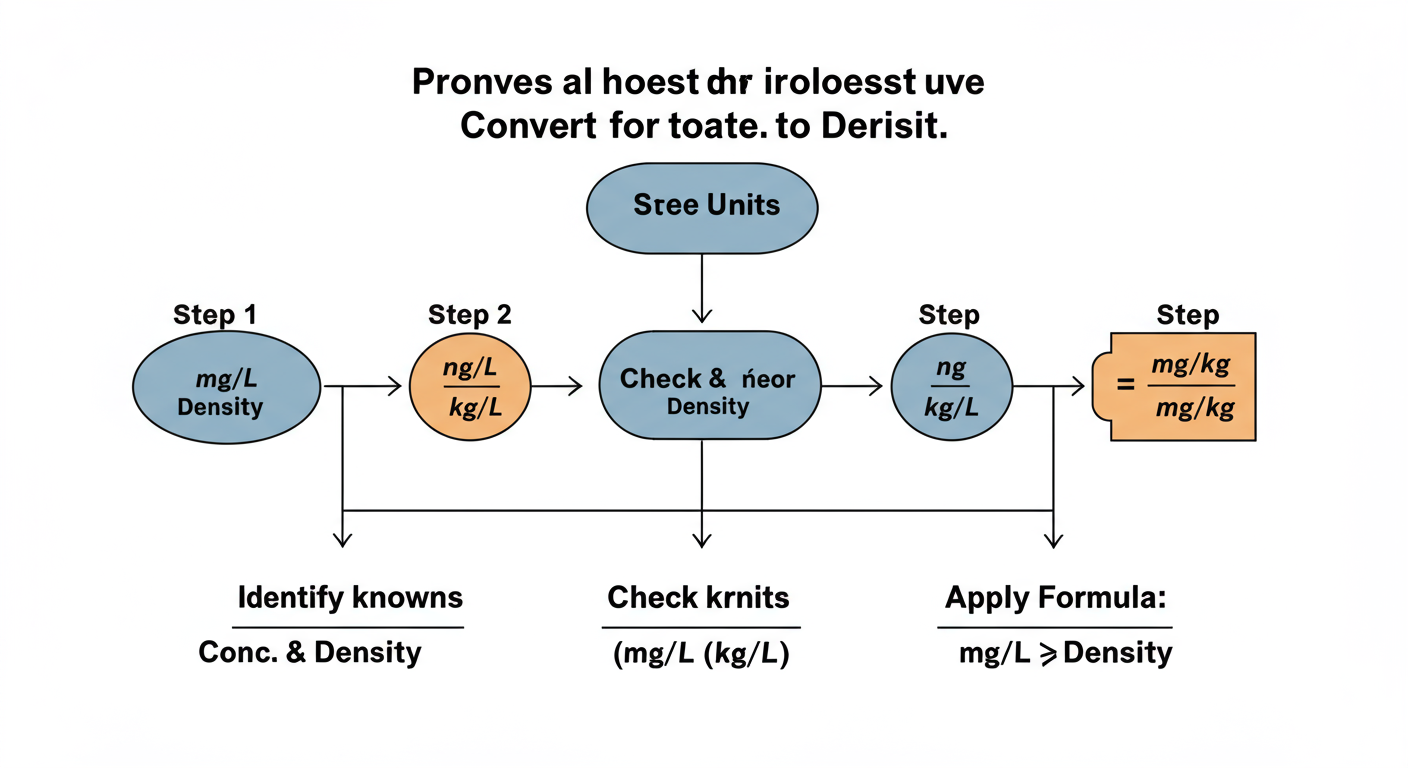
Step 1: Find Your Known Values
First, you need two pieces of information:
- The concentration in mg/L. This is your starting point, usually from a lab report or measurement.
- The density of the liquid/solution. This is the crucial part. Is it a dilute water solution where you can assume 1.0 kg/L? Or is it something else? For anything other than dilute water, you MUST find the specific density.
💡 Pro Tip
Where do you find density? Check the substance’s Safety Data Sheet (SDS), which is required for most chemicals. You can also consult engineering resources like Perry’s Chemical Engineers’ Handbook or reliable online databases. Always note the temperature at which the density was measured, as it can change. For an authoritative look at how temperature affects water density, the USGS Water Science School is an excellent resource.
Step 2: Ensure Your Units Are Consistent
Before you touch a calculator, check your units. The formula works perfectly if you have:
- Concentration in mg/L
- Density in kg/L
What if your density is in grams per milliliter (g/mL) or grams per cubic centimeter (g/cm³)? You’re in luck. 1 g/mL = 1 kg/L, so you can use the number directly. If it’s in any other unit, you must convert it to kg/L first.
Step 3: Apply the Formula and Calculate
Now for the easy part. Just plug your values into the formula:
mg/kg = (Your concentration in mg/L) / (The solution’s density in kg/L)
The number you get is your accurate concentration in mg/kg. Done.
🎯 Key Takeaway
To convert mg/L to mg/kg, you must divide the concentration in mg/L by the solution’s density in kg/L. Forgetting density is the single biggest mistake you can make. For dilute water, you can often use a density of 1.0 kg/L, but for any other substance, you must find its specific density.
Real-World Scenarios: Putting the Formula to the Test
Let’s see how this plays out in the real world. The difference between getting it right and wrong can be massive.
Imagine a lab reports a 100 mg/L concentration of a chemical. Let’s see how the final mg/kg value changes based on the substance.
| Substance | Density (approx. kg/L) | Calculation (100 mg/L ÷ Density) | Result (mg/kg) |
|---|---|---|---|
| Dilute Wastewater | 1.0 kg/L | 100 / 1.0 | 100 mg/kg |
| Seawater Brine | 1.2 kg/L | 100 / 1.2 | 83.3 mg/kg |
| Ethanol | 0.79 kg/L | 100 / 0.79 | 126.6 mg/kg |
| Contaminated Mud Slurry | 1.5 kg/L | 100 / 1.5 | 66.7 mg/kg |
Look at those results. The same 100 mg/L concentration can mean anything from 66.7 mg/kg to 126.6 mg/kg. If a regulatory limit was 100 mg/kg, using the wrong density could be the difference between compliance and a hefty fine.
Avoiding Critical Errors: Common Mistakes and How to Fix Them
In our experience reviewing technical reports, we see the same few mistakes over and over. Here’s how to avoid them.
⚠️ Watch Out: Reversing the Formula
A surprisingly common error is multiplying by density instead of dividing. Remember, you want to cancel out the volume unit (Liters). The only way to do that is by dividing mg/L by kg/L. Multiplying will give you a nonsensical unit and a wildly incorrect answer.
Mistake #1: Using the Water Shortcut for Everything
As we’ve hammered home, assuming a density of 1.0 kg/L for anything that isn’t dilute water is a critical error.
The Fix: Always ask, “Is this solution basically water?” If the answer is no, or even “I’m not sure,” take the extra two minutes to find the specific density. It’s worth it.
Mistake #2: Ignoring Temperature
Density isn’t a fixed constant; it changes with temperature. For most liquids, density decreases as temperature increases. While this effect is minor for water under normal conditions, it can be significant for organic solvents or in industrial processes with wide temperature swings.
The Fix: When you look up a density value, check the temperature it was measured at. If your process operates at a very different temperature, you may need to find a temperature-corrected density value for high-precision work.
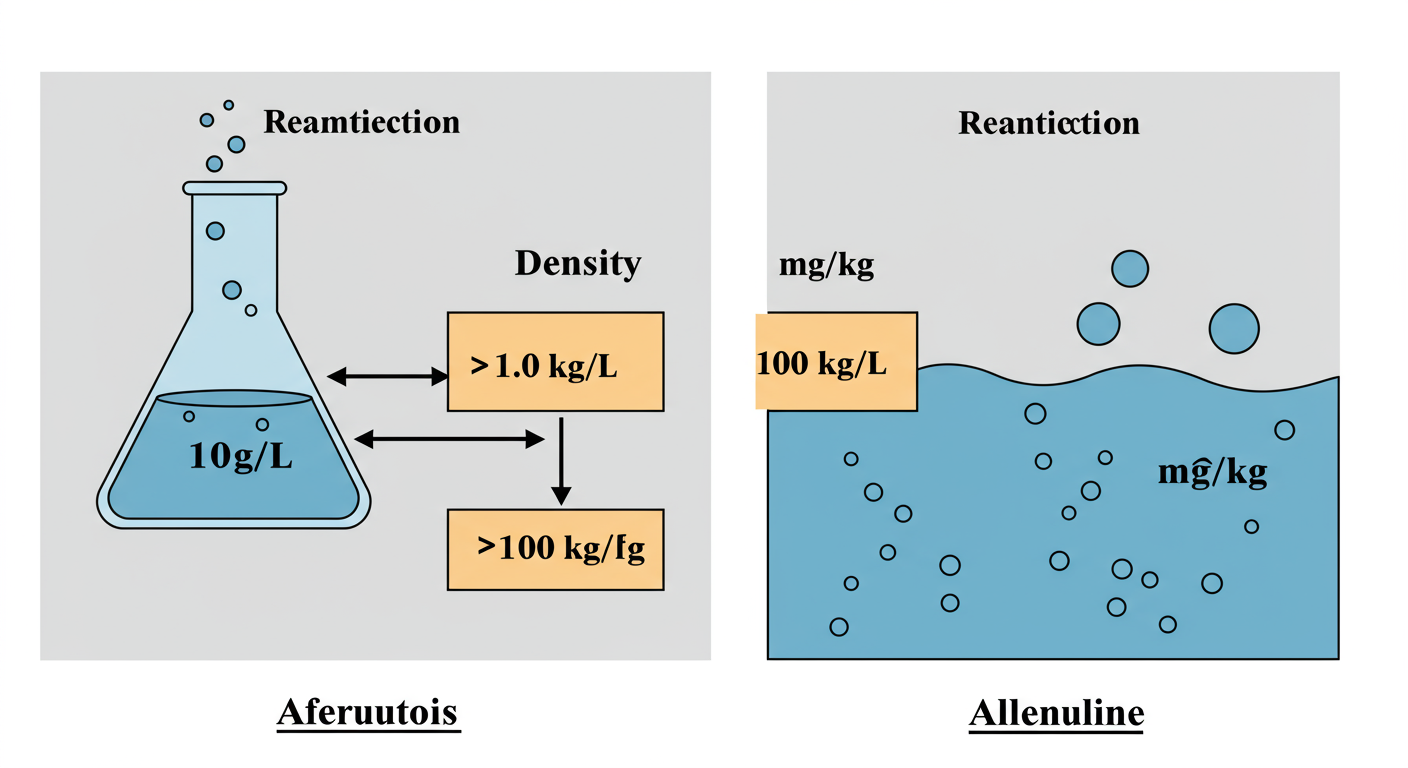
💡 Pro Tip: The “Sanity Check”
After you calculate, perform a quick sanity check.
- If your liquid is denser than water (>1.0 kg/L), your final mg/kg value should be lower than the starting mg/L value.
- If your liquid is less dense than water (<1.0 kg/L), your final mg/kg value should be higher than the starting mg/L value.
This simple check catches most calculation errors, like accidentally multiplying instead of dividing.
❓ Frequently Asked Questions
Is mg/L exactly the same as ppm?
For dilute aqueous solutions, yes, mg/L is effectively the same as parts per million (ppm). This is because 1 Liter of water weighs 1 kilogram (1,000,000 milligrams), so 1 mg in 1 L is one part in a million. This approximation falls apart for any liquid whose density isn’t 1.0 kg/L.
What if I need to convert mg/kg back to mg/L?
You just reverse the formula! To go from a mass-based concentration back to a volume-based one, you multiply. The formula is: Concentration (mg/L) = Concentration (mg/kg) × Density of the Solution (kg/L).
Where can I find the density of soil or sediment?
This is trickier, as soil density (called bulk density) varies widely with composition, compaction, and water content. It’s not a fixed value you can look up. For accurate environmental work, a sample of the soil or sediment must be collected and its bulk density measured in a lab. Typical values range from 1.1 to 1.6 kg/L.
Does pressure affect density?
For liquids, the effect of pressure on density is very small under normal atmospheric conditions and can usually be ignored. For gases, pressure has a massive effect on density, but the mg/L to mg/kg conversion is less common for gases anyway. For most practical applications discussed here, you only need to worry about temperature.
What are some other common concentration units?
In scientific and technical fields, you’ll see many units. Besides mg/L and mg/kg, you might encounter micrograms per liter (µg/L), molarity (moles/L), and normality (equivalents/L). Each has a specific use, and converting between them requires understanding the underlying principles, much like the conversion we’ve discussed here. For a great overview of measurement units in chemistry, the Chemistry LibreTexts project is a fantastic educational resource.
Conclusion: From Volume to Mass with Total Confidence
The journey from mg/L to mg/kg isn’t about memorizing a formula. It’s about understanding the fundamental difference between volume and mass—and knowing that density is the only reliable bridge between them.
You now have the knowledge that separates casual guesswork from professional accuracy. You know the 1:1 shortcut is a handy tool for dilute water, but you also know its dangerous limitations. You have a bulletproof, 3-step process that works for any liquid, from wastewater to industrial solvents.
So the next time you’re faced with a lab report, don’t just guess. Find the density. Do the division. Perform the sanity check. You’ll have an answer that is not only numerically correct but scientifically sound and defensible. That’s not just good math; it’s good science.



