Ever stared at a lab report, a value in mg/L in one hand and a regulatory limit in mg/kg in the other, and felt a wave of uncertainty? You’re not alone. It’s a common scenario in environmental science, agriculture, and chemistry, and a simple mistake here can lead to costly compliance issues or flawed research.
Here’s the hard truth: you can’t just swap the units. A liter of industrial sludge doesn’t weigh the same as a liter of water. Assuming it does is a recipe for disaster.
But don’t worry. This isn’t some arcane chemical magic. It’s a straightforward process that hinges on one single, crucial factor. In this guide, we’ll break down exactly how to convert mg/L to mg/kg accurately, every single time. You’ll walk away with the formula, real-world examples, and the confidence to know your numbers are rock-solid.
📑 What You’ll Learn
Why You Can’t Just Swap mg/L for mg/kg
Let’s get this out of the way first. The biggest misconception we see is treating mg/L and mg/kg as interchangeable. This only works in one very specific, idealized situation: pure water at 4°C, where its density is almost exactly 1.0 kg/L. In that perfect world, one liter weighs one kilogram, so the units effectively cancel out.
But we don’t work in a perfect world. We work with soil slurries, wastewater, chemical solutions, and brines. A liter of honey is much heavier than a liter of gasoline. That’s the entire problem in a nutshell.
You’re dealing with two fundamentally different types of measurement:
- mg/L (milligrams per liter) is a measure of mass per volume. It asks: “How much stuff (in mg) is in a certain amount of space (a liter)?”
- mg/kg (milligrams per kilogram) is a measure of mass per mass. It asks: “How much stuff (in mg) is in a certain amount of weight (a kilogram)?”
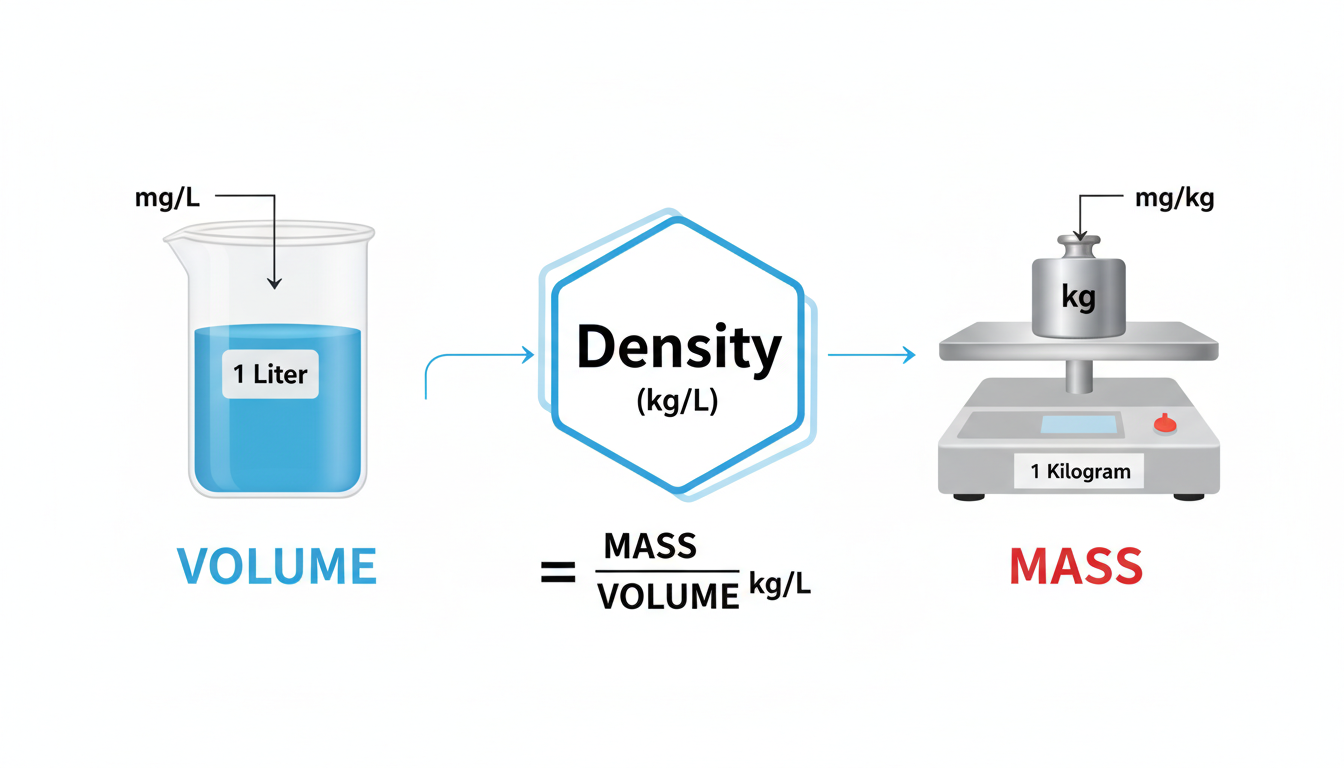
To bridge the gap between volume (space) and mass (weight), you need the secret ingredient. You need density.
| Metric | mg/L (Milligrams per Liter) | mg/kg (Milligrams per Kilogram) |
|---|---|---|
| Measurement Type | Mass / Volume | Mass / Mass |
| Commonly Used For | Liquids, aqueous solutions, drinking water analysis | Solids (soil, sediment), sludges, tissues, sometimes liquids |
| Also Known As | Parts per million (ppm) only in dilute water | Parts per million (ppm) by mass |
| Key Dependency | The volume of the liquid | The mass of the substance |
Density: The One Factor You Absolutely Can’t Ignore
Think of density as the “heaviness” of a substance packed into a specific space. Its formula is simple: Density (ρ) = Mass (m) / Volume (V). For our purposes, the most useful unit for density is kilograms per liter (kg/L).
When you want to convert mg/L to mg/kg, you’re really asking: “Okay, I have a concentration in one liter, but how much does that liter actually weigh?”
- If a liter of your substance weighs more than 1 kg (like a soil slurry at 1.5 kg/L), the same amount of contaminant is spread over a larger mass. Your mg/kg value will be lower than your mg/L value.
- If a liter of your substance weighs less than 1 kg (like gasoline at 0.75 kg/L), the same amount of contaminant is concentrated in a smaller mass. Your mg/kg value will be higher than your mg/L value.
It’s an inverse relationship. And it makes all the difference.
💡 Pro Tip
Don’t have a lab-measured density? You’re not out of luck. For common substances, you can often find reliable density values in engineering handbooks or online databases. Authoritative sources like the Wikipedia page on Density provide tables for many materials. Just be sure the temperature and conditions match your sample as closely as possible.
The 5-Step Formula to Convert mg/L to mg/kg
Ready for the math? It’s simpler than you think. The entire conversion boils down to one elegant formula.
Concentration (mg/kg) = Concentration (mg/L) / Density (kg/L)
That’s it. Let’s walk through it step-by-step with a practical example. Imagine you’re analyzing industrial wastewater for a copper contaminant.
Step 1: Get Your Starting Value (mg/L)
Your lab report comes back. The sample contains 150 mg/L of copper. This is your starting point.
Step 2: Find the Density (kg/L)
This is the most crucial step. You can’t guess. You measure the density of the wastewater and find it’s 1.15 kg/L. It’s slightly heavier than pure water due to dissolved solids and chemicals.
Step 3: Check Your Units
Before you calculate, do a quick sanity check. Is your concentration in mg/L? Yes. Is your density in kg/L? Yes. You’re good to go. If your density was in g/mL or g/cm³, you’re also fine, as 1 g/mL = 1 kg/L. Any other unit needs to be converted first.
Step 4: Plug and Chug
Now, apply the formula:
Concentration (mg/kg) = 150 mg/L / 1.15 kg/L
Concentration (mg/kg) ≈ 130.43 mg/kg
Step 5: State Your Final Answer
The mass-based concentration of copper in the wastewater is 130.43 mg/kg. Notice how it’s lower than the 150 mg/L value? That’s because each liter of the wastewater weighs more than a kilogram, effectively diluting the concentration when measured by mass.

⚠️ Watch Out
A simple slip-up with units can invalidate your entire calculation. If a datasheet gives you density in kg/m³, remember that there are 1,000 liters in a cubic meter (m³). To convert kg/m³ to kg/L, you must divide by 1,000. Forgetting this step will throw your result off by a factor of a thousand. Always double-check.
Real-World Scenarios: Where This Conversion is Critical
This isn’t just a textbook exercise. From our experience analyzing client data, getting this conversion right has major real-world consequences.
Scenario 1: Environmental Soil Remediation
An environmental consultant is assessing a contaminated site. A lab mixes a soil sample with water to create a slurry, then tests the liquid portion (leachate). The test finds 80 mg/L of lead in the leachate. The density of the slurry is measured at 1.6 kg/L.
- The Wrong Way: Reporting 80 mg/kg, assuming a 1:1 conversion.
- The Right Way: 80 mg/L / 1.6 kg/L = 50 mg/kg.
That’s a 37.5% difference! This could be the difference between a site requiring millions of dollars in cleanup and one that is deemed safe. For official guidance, professionals often refer to standards set by bodies like the U.S. Environmental Protection Agency (EPA).
Scenario 2: Agriculture & Fertigation
A farmer is applying a liquid fertilizer through an irrigation system. The fertilizer solution has a nutrient concentration of 500 mg/L and a density of 1.1 kg/L. They need to calculate the application rate based on the mass of nutrients being applied to the soil.
- Calculation: 500 mg/L / 1.1 kg/L = 454.5 mg/kg.
This tells the farmer exactly how much nutrient mass is in each kilogram of the solution they are applying, allowing for precise and efficient fertilization without waste.
💡 Pro Tip
What about Parts Per Million (PPM)? For very dilute solutions in water (like tap water analysis), you can safely assume that 1 mg/L is equal to 1 ppm. This is because the tiny amount of dissolved substance doesn’t significantly change the density from 1.0 kg/L. But the moment you’re dealing with anything else—brines, sludges, industrial effluents—that shortcut goes out the window. Stick to the density formula.
🎯 Key Takeaway
The conversion from mg/L to mg/kg is not a direct unit swap; it is a calculation that is entirely dependent on the density of your substance. The core formula is simple: divide the concentration in mg/L by the density in kg/L. Ignoring density is the single most common and costly mistake you can make.
Avoiding the Costly Mistakes Everyone Else Makes
We’ve seen the same errors trip people up time and time again. Here’s what to watch for.
The “Dry Weight” vs. “Wet Weight” Trap
When analyzing soil, sediment, or sludge, results are often required on a “dry weight” basis. Your initial calculation gives you the concentration in the wet, as-is sample. To get the dry weight concentration, you must also know the moisture content of the sample and adjust your calculation accordingly. This is a critical extra step in geotechnical and environmental fields.
⚠️ Watch Out
Never assume a “standard” density for a critical application. The density of soil can range from 1.1 kg/L for loose topsoil to over 1.6 kg/L for compacted clay. Using a generic value of 1.3 kg/L when the actual value is 1.6 kg/L can introduce a 20% error into your final result. When precision matters, always use a measured density for your specific sample.
Forgetting About Temperature
Does temperature matter? For most routine applications, probably not enough to worry about. But for high-precision scientific or calibration work, it absolutely does. The density of liquids changes with temperature. Water, for example, is most dense at 4°C (1.000 kg/L) but becomes less dense as it warms up or cools down toward freezing. Industry resources like The Engineering ToolBox provide detailed tables showing these variations.
| Substance | Typical Density (kg/L) | Calculation | Resulting Concentration (mg/kg) |
|---|---|---|---|
| Gasoline | 0.75 | 100 / 0.75 | 133.3 mg/kg (Higher) |
| Pure Water | 1.00 | 100 / 1.00 | 100.0 mg/kg (Same) |
| Seawater | 1.03 | 100 / 1.03 | 97.1 mg/kg (Lower) |
| Heavy Sludge | 1.40 | 100 / 1.40 | 71.4 mg/kg (Much Lower) |
Conclusion: From Confusion to Confidence
Mastering the conversion from mg/L to mg/kg elevates you from someone who just reads numbers to someone who truly understands them. It’s not about memorizing a complex process; it’s about internalizing one simple truth: it’s all about density.
By understanding the fundamental difference between mass-based and volume-based concentrations, and by consistently applying the simple formula—mg/kg = mg/L ÷ density—you can ensure your data is accurate, defensible, and meaningful. The next time you see those units on a report, you won’t feel uncertainty. You’ll feel confident, because you’ll know exactly what to do.
Your next step? The next time you have a sample, don’t just ask for the concentration. Ask for the density, too. It’s the key to unlocking the right answer.
❓ Frequently Asked Questions
Is mg/L the same as ppm?
Only for dilute aqueous solutions where the density is very close to 1.0 kg/L. In this specific case, 1 mg/L ≈ 1 ppm. For any other substance (oil, sludge, concentrated solutions), this direct equivalence is incorrect and you must use the density-based formula.
What is the direct formula to convert mg/L to mg/kg?
The formula is: Concentration (mg/kg) = Concentration (mg/L) / Density (in kg/L). You absolutely must know the density of the substance to perform an accurate conversion.
How do I convert mg/kg back to mg/L?
Easy. You just rearrange the formula: Concentration (mg/L) = Concentration (mg/kg) × Density (in kg/L). As always, having an accurate density value is non-negotiable.
Why is density so important for this conversion?
Because density is the physical property that connects a substance’s volume (Liters) to its mass (Kilograms). Since mg/L is a mass/volume unit and mg/kg is a mass/mass unit, density is the essential bridge that allows you to translate between the two.
What’s a typical density for soil?
It varies widely. The bulk density of dry soil typically ranges from 1.1 kg/L for loose, organic soils to 1.6 kg/L for compacted mineral soils. A soil slurry (soil mixed with water) will have a different density, often in the 1.3 to 1.8 kg/L range, depending on the soil-to-water ratio.
Can I use an online calculator for this?
You can, but be careful. A good calculator will always require you to input the density. If a calculator offers to convert mg/L to mg/kg without asking for a density value, it is making a dangerous assumption (that density is 1.0 kg/L) and should not be trusted for anything other than pure water.



