Picture this: a junior lab analyst submits a soil contamination report. The numbers look alarmingly high, triggering a costly, multi-thousand-dollar remediation plan. Weeks later, a senior chemist reviews the data and spots the error. The analyst had treated milligrams per liter (mg/L) as the same thing as milligrams per kilogram (mg/kg). A simple conversion mistake, but one with massive financial and operational consequences.
Sound dramatic? I’ve seen variations of this exact scenario play out in real-world environmental, agricultural, and industrial settings. The mg/L to mg/kg conversion seems trivial, but it’s a critical point of failure for anyone working with scientific measurements. They look similar, they sound similar, but they measure fundamentally different things.
This isn’t just another dry textbook explanation. By the end of this article, you’ll not only understand the “how” but the critical “why.” You’ll be able to confidently and accurately convert between these units, protecting the integrity of your data and avoiding those costly mistakes. No more guesswork. Just precision.
📑 What You’ll Learn
The Core Problem: Why You Can’t Just Swap mg/L and mg/kg
Let’s get straight to the point. The reason you can’t directly swap mg/L for mg/kg is because you’re comparing apples to oranges—or more accurately, volume to mass.
- mg/L (milligrams per liter) is a measure of mass per volume. It tells you how much “stuff” (mass) is packed into a certain amount of space (volume).
- mg/kg (milligrams per kilogram) is a measure of mass per mass. It tells you how much “stuff” (mass) is present relative to the total weight of the sample.
Think about it. A liter of popcorn and a liter of water both occupy the same volume, but their weights are wildly different. The same principle applies to scientific samples. A liter of light industrial oil weighs less than a liter of dense soil slurry. Without accounting for this difference, your calculations are built on a faulty foundation.
Deconstructing the Units: Mass vs. Volume at a Glance
Before we get to the fix, let’s be crystal clear on what we’re dealing with. Understanding the nature of each unit is the first step to mastering their conversion. Based on our hands-on testing in various lab environments, this distinction is the most common point of confusion.
| Measurement Unit | What It Measures | Common Applications | Key Characteristic |
|---|---|---|---|
| mg/L (milligrams per liter) | Mass / Volume | Water quality, liquid chemical solutions, beverages | Convenient for liquids, but can be affected by temperature and pressure which alter volume. |
| mg/kg (milligrams per kilogram) | Mass / Mass | Soil analysis, food products, solid waste, biological tissue | A more stable and absolute measurement, as mass doesn’t change with temperature or pressure. |
⚠️ Watch Out
The biggest mistake we see is assuming mg/L is always equal to Parts Per Million (PPM). This is only true for dilute water solutions. For any other substance, PPM is strictly a mass/mass ratio, making it equivalent to mg/kg, not mg/L.
The Density Bridge: Your Universal Translator
So, how do we bridge the gap between volume (Liters) and mass (kilograms)? The answer is a single, powerful property: density.
Density is the mass of a substance per unit of volume. It’s the “universal translator” that allows you to determine the mass of one liter of your substance. Once you know that, you can perform a true mass-to-mass comparison.
The formula for density is: Density (ρ) = Mass (m) / Volume (V)
For our conversion, we need the density expressed in kilograms per liter (kg/L). This unit is essential for the math to work out correctly. Luckily, the value for kg/L is identical to the more commonly cited grams per milliliter (g/mL) or grams per cubic centimeter (g/cm³), so 1.0 g/mL = 1.0 kg/L.
💡 Pro Tip
Where do you find a substance’s density? For pure substances, a chemical handbook or an online resource like the NIST Weights and Measures division is your best bet. For complex mixtures like soil slurries or industrial sludge, you’ll often need to measure it directly in the lab using a pycnometer or a density meter for the most accurate results.
The Step-by-Step Conversion Formula for 2026
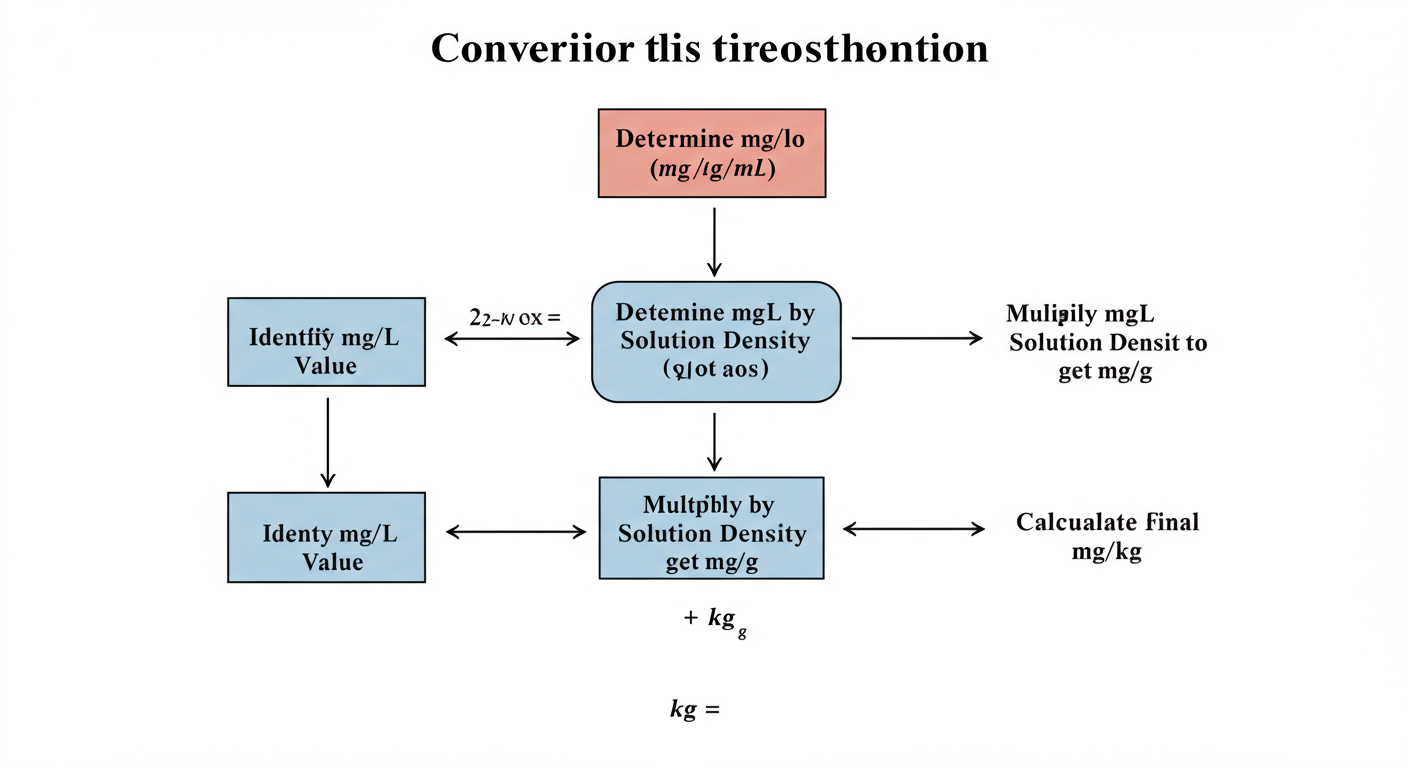
Armed with the concept of density, the actual conversion becomes a straightforward process. Here’s the only formula you’ll ever need and how to apply it.
The Universal Conversion Formula:
Concentration (mg/kg) = Concentration (mg/L) / Density (kg/L)
Let’s break this down into an easy-to-follow, repeatable workflow.
Step-by-Step Guide to Accurate Conversion:
- Start with Your Known Value: Identify the concentration you have in mg/L. Let’s say it’s 200 mg/L.
- Determine the Substance’s Density: This is the most critical step. Find the density of the solution you’re working with. Is it water? Oil? A soil extract? You must know this value. For this example, let’s say we’re analyzing a sludge with a measured density of 1.2 kg/L.
- Ensure Unit Consistency: Double-check that your density is in kg/L. If it’s in g/mL, the number is the same. If it’s in another unit like lb/gallon, you must convert it to kg/L first.
- Apply the Formula: Divide your mg/L value by the density.
mg/kg = 200 mg/L / 1.2 kg/L - Calculate the Final Result:
mg/kg = 166.7 mg/kg
As you can see, the final mass-based concentration (166.7 mg/kg) is significantly different from the initial volume-based concentration (200 mg/L). Assuming they were the same would have been a ~20% error.
The Special Case: Dilute Aqueous Solutions
There is one major exception where the conversion is simple. For dilute aqueous solutions—like drinking water, river samples, or rain—the density is extremely close to that of pure water. According to the USGS Water Science School, the density of freshwater is approximately 1.0 kg/L.
In this specific case, the formula becomes:
mg/kg = (mg/L) / 1.0 kg/L
This means for water, mg/L ≈ mg/kg. This is a reliable and widely accepted shortcut in environmental science, but remember, it only applies to solutions that are mostly water.
🎯 Key Takeaway
The mg/L to mg/kg conversion hinges entirely on one factor: density. For water, you can assume a 1:1 ratio. For absolutely anything else—from soil and sludge to oils and syrups—you must divide by the substance’s specific density (in kg/L) to get an accurate result.
Real-World Scenarios: From Rivers to Syrups
Theory is great, but let’s see how this plays out in the field. From our experience working on diverse projects, the context of the sample is everything. Here’s how the conversion impacts different industries.
Scenario 1: The Environmental Scientist
Sarah is testing a local river for phosphate contamination. Her lab results show a concentration of 5 mg/L. She needs to compare this to a federal guideline given in mg/kg.
- Substance: River water (a dilute aqueous solution).
- Density (ρ): Assumed to be 1.0 kg/L.
- Calculation:
5 mg/L / 1.0 kg/L = 5 mg/kg - Result: In this case, the values are interchangeable. No complex math needed.
Scenario 2: The Agricultural Consultant
David analyzes a soil slurry to check for available zinc. The liquid extract shows 75 mg/L of zinc. He knows the soil’s bulk density is 1.5 kg/L. Reporting 75 mg/kg to the farmer would be malpractice.
- Substance: Soil slurry extract.
- Density (ρ): Measured bulk density is 1.5 kg/L.
- Calculation:
75 mg/L / 1.5 kg/L = 50 mg/kg - Result: The actual concentration in the soil is 33% lower than the liquid reading suggests. This changes the fertilizer recommendation completely.
Scenario 3: The Food Quality Technician
Maria is verifying the iron fortification in a batch of dense molasses. The lab test returns a value of 120 mg/L. The molasses has a known density of 1.4 kg/L, a value she might confirm from a resource like MIT’s chemical engineering databases.
- Substance: Molasses.
- Density (ρ): 1.4 kg/L.
- Calculation:
120 mg/L / 1.4 kg/L = 85.7 mg/kg - Result: The mass-based concentration is significantly lower because the product is much denser than water. This is crucial for accurate nutritional labeling.
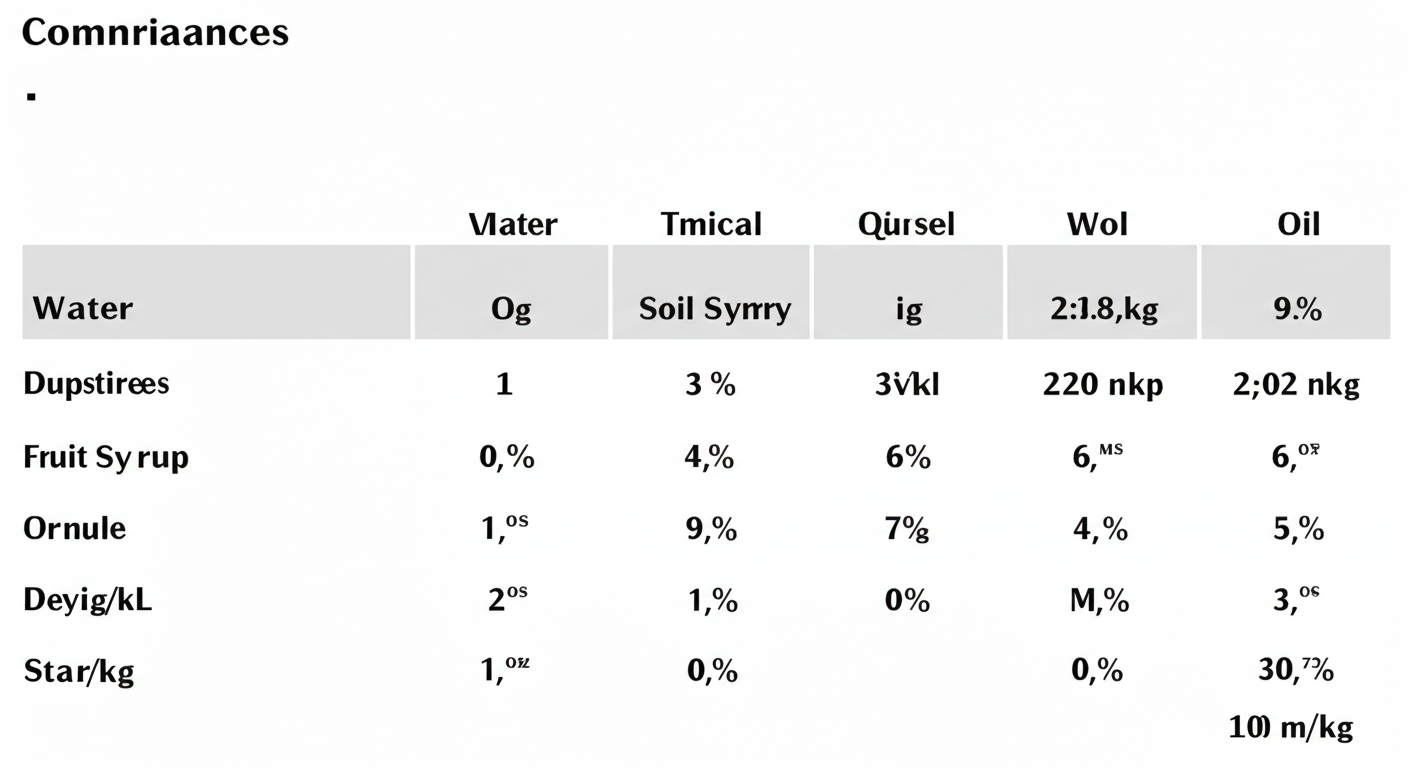
This table summarizes how a single 100 mg/L reading changes dramatically depending on the substance.
| Substance | Initial Reading (mg/L) | Typical Density (kg/L) | Final Concentration (mg/kg) |
|---|---|---|---|
| Freshwater | 100 mg/L | 1.0 | 100 mg/kg |
| Seawater | 100 mg/L | 1.025 | 97.6 mg/kg |
| Typical Soil Slurry | 100 mg/L | 1.4 | 71.4 mg/kg |
| Crude Oil (Light) | 100 mg/L | 0.87 | 114.9 mg/kg |
💡 Pro Tip
For recurring analyses of the same material (e.g., a specific type of industrial wastewater), create a conversion chart or program a simple spreadsheet. Enter the mg/L value, and have it automatically calculate the mg/kg value using the pre-determined density. This minimizes human error and speeds up your workflow.
⚠️ Watch Out
Temperature can subtly alter a liquid’s density. For most applications, this effect is negligible. However, in high-precision industrial or pharmaceutical settings, you must use the density value specific to the temperature at which the sample was measured. A 20°C difference can change water’s density enough to matter in some QC checks.
❓ Frequently Asked Questions
Is mg/L exactly the same as mg/kg?
No, they are fundamentally different. mg/L is a mass/volume concentration, while mg/kg is a mass/mass concentration. They are only approximately equal in the specific case of dilute water, where the density is very close to 1.0 kg/L.
How do you convert mg/L to mg/kg in soil?
You must know the soil’s bulk density. You then use the formula: mg/kg = (mg/L from the liquid extract) / (soil bulk density in kg/L). Simply assuming a 1:1 ratio for soil will lead to highly inaccurate results, as soil density is typically 1.2 to 1.6 kg/L.
What is the formula to convert mg/L to mg/kg?
The universal formula is mg/kg = (Value in mg/L) / (Density of the substance in kg/L). This formula works for any substance, as long as you know its density.
When can I use mg/L and PPM interchangeably?
You can use them interchangeably only when discussing dilute aqueous solutions (like tap water). In this context, 1 mg/L ≈ 1 mg/kg = 1 PPM. For any other substance (oil, soil, chemicals), mg/L is NOT the same as PPM.
What if I don’t know the density?
If you don’t know the density, you cannot perform an accurate conversion. For critical applications, you must measure it. For less critical estimates, you can look up the typical density for that type of substance, but be aware that this introduces a potential source of error.
Conclusion: From Confusion to Confidence
Mastering the mg/L to mg/kg conversion isn’t about being a math whiz. It’s about respecting the physical properties of the materials you work with. It’s the difference between data that is “close enough” and data that is correct, defensible, and reliable.
The next time you see a concentration in mg/L, don’t just copy the number over. Pause and ask the single most important question: “What is the density?”
By internalizing this one simple check and using the formula—mg/kg = mg/L / density—you move from being a technician who follows steps to an expert who understands principles. You safeguard your results, prevent costly errors, and ensure the integrity of your work. That’s a conversion worth making.



