Here’s the thing about water quality reports: they are terrifyingly precise. One decimal point in the wrong place can mean the difference between “safe to drink” and a federally mandated shutdown.
If you’ve ever stared at a lab report wondering why one column says μg/L and the regulatory limit is listed in ppb, you aren’t alone. It’s a frustration I’ve seen seasoned lab technicians and worried homeowners face alike. In 2026, with environmental regulations becoming stricter than ever, understanding these units isn’t just academic—it’s a safety requirement.
The good news? For most water-based applications, the conversion of micrograms per liter to ppb is incredibly simple. The bad news? If you apply that simple rule to the wrong liquid—like seawater or industrial solvents—your data will be dead wrong.
In this guide, we’re going to cut through the academic noise. We’ll look at the real-world math, the dangerous exceptions, and exactly how to interpret your data like a pro.
📑 What You’ll Learn
The Golden Rule: Micrograms Per Liter to PPB Explained
Let’s start with the answer you came here for. If you are dealing with fresh water at standard temperatures, the conversion is a direct 1:1 swap.
1 Microgram per Liter (μg/L) = 1 Part Per Billion (ppb)
That’s it. If your lab report says the arsenic level is 5 μg/L, that is exactly 5 ppb. You don’t need a calculator, and you don’t need a degree in stoichiometry.
But why?
In my years working with environmental data, I’ve found that people memorize this rule but don’t understand the mechanics, which leads to mistakes later. It works because of a convenient coincidence in the metric system regarding water.
🎯 Key Takeaway
For fresh water, 1 μg/L is exactly 1 ppb. This works because 1 liter of water weighs exactly 1 kilogram (1 billion micrograms). If you are testing water, simply swap the units.
The Science: Why Density Dictates the Math
To understand converting micrograms per liter to ppb, you have to look at what the units actually measure.
- Micrograms per Liter (μg/L): This is a weight-to-volume ratio. It tells you how much mass of a contaminant is floating in a specific volume of liquid.
- Parts Per Billion (ppb): This is usually a weight-to-weight ratio. It tells you how many units of mass (contaminant) are in one billion units of mass (total solution).
The bridge between volume (Liters) and weight (Kilograms) is density.
Water has a density of approximately 1 kg/L. Since 1 kilogram equals 1,000,000,000 (one billion) micrograms, having 1 microgram in that liter is literally one part in a billion.
Visualizing the Scale: PPM vs. PPB
It’s easy to get lost in the zeroes. When we talk about trace contaminants, we are talking about incredibly small amounts. Yet, in toxicology, these small amounts matter immensely.
Here is how the hierarchy of units breaks down. I’ve laid this out to help you visualize the difference between “Million” and “Billion.”
| Unit | Abbreviation | Metric Equivalent (in Water) | Real-World Analogy |
|---|---|---|---|
| Parts Per Million | ppm | 1 mg/L (Milligram/Liter) | 1 drop of water in a fuel tank of a compact car. |
| Parts Per Billion | ppb | 1 μg/L (Microgram/Liter) | 1 drop of water in a full Olympic-sized swimming pool. |
| Parts Per Trillion | ppt | 1 ng/L (Nanogram/Liter) | 1 drop of water in 20 Olympic-sized swimming pools. |
💡 Pro Tip
Always double-check the prefix! A common data entry error is confusing mg/L (milligrams) with μg/L (micrograms). This is a factor of 1,000. Mistaking 5 ppm for 5 ppb could mean missing a lethal dose of contamination.
The Density Trap: When the Rule Breaks
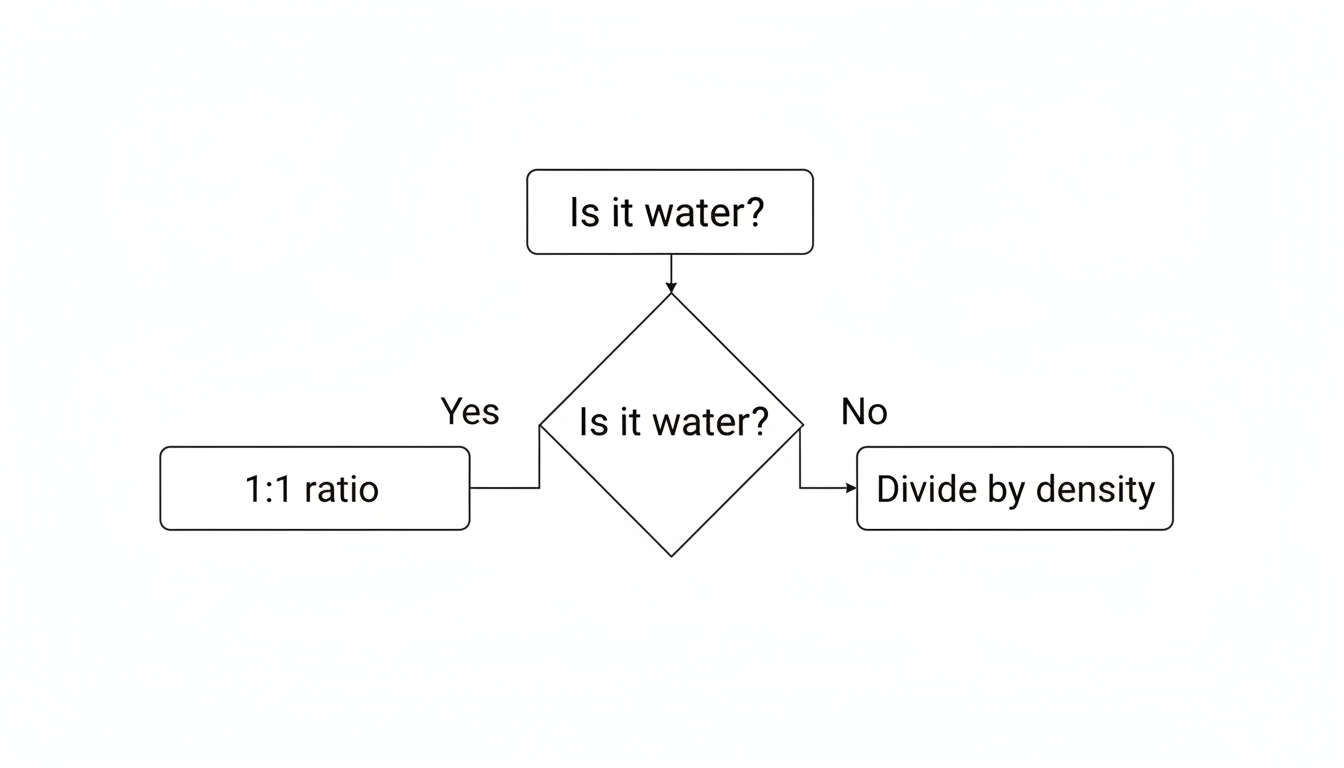
Here is where the “expert” part of this guide kicks in. The 1:1 rule assumes the liquid you are testing has a density of 1.0 kg/L.
But what if you aren’t testing tap water? What if you are analyzing industrial wastewater, seawater, or a chemical solvent?
If the liquid is denser than water, a liter of it weighs more than 1 kg. This throws off the “parts per billion” calculation because the “billion” parts (the total solution) are heavier.
The Universal Formula:
$$ text{Concentration (ppb)} = frac{text{Concentration } (mu g/L)}{text{Density of Solution (kg/L)}} $$
Let’s look at how this plays out in different scenarios:
| Substance | Approx. Density (kg/L) | Lab Reading | True Concentration (ppb) |
|---|---|---|---|
| Fresh Water | 1.00 | 100 μg/L | 100 ppb |
| Seawater | 1.025 | 100 μg/L | 97.5 ppb |
| Sulfuric Acid (Conc.) | 1.84 | 100 μg/L | 54.3 ppb |
| Gasoline | 0.74 | 100 μg/L | 135.1 ppb |
Look at the gasoline example. Because gasoline is lighter than water, 100 μg/L actually represents a higher concentration in ppb (135 ppb). If you used the 1:1 water rule here, you would significantly underreport the contamination level.

⚠️ Watch Out
Do NOT use this conversion for gases. Converting μg/m³ to ppb in air is a completely different physical process that requires the Ideal Gas Law, temperature, and molecular weight. The 1:1 rule is strictly for liquid water.
Step-by-Step Conversion Guide
Ready to convert your data? Follow this workflow to ensure 100% accuracy every time.
- Identify the Solute and Solvent: Confirm you are looking at a contaminant (solute) dissolved in a liquid (solvent).
- Check the Units: Ensure your starting value is in μg/L. If it is in mg/L, multiply by 1,000 first.
- Determine Density:
- Is it fresh water? Assume Density = 1 kg/L.
- Is it wastewater, brine, or a chemical? Measure the specific gravity or consult a reference like PubChem.
- Calculate: Divide your μg/L value by the density.
- Verify Compliance: Compare your final ppb figure against the regulatory limit.
Real-World EPA Limits (2026 Context)
Why do we obsess over converting micrograms per liter to ppb? Because the US Environmental Protection Agency (EPA) and other global bodies set their Maximum Contaminant Levels (MCLs) using these units interchangeably.
In 2026, we’ve seen tightened regulations on PFAS (forever chemicals) and heavy metals. Here is a snapshot of current critical limits where this conversion is vital:
- Lead: The action level is 15 ppb. If your lab report reads 16 μg/L, you are non-compliant.
- Arsenic: The federal limit is 10 ppb. A reading of 10 μg/L is right on the threshold.
- PFAS (PFOA/PFOS): These are now regulated in the ppt (parts per trillion) range. If you see a report of 0.004 μg/L, you must convert that to 4 ppt (parts per trillion) to understand the severity.
⚠️ Watch Out
Temperature Matters (Slightly). Water expands when it gets hot. At 90°C, water density drops to about 0.965 kg/L. While usually negligible for home testing, in high-precision industrial chemistry, testing hot wastewater without correcting for density can skew your ppb results by nearly 4%.
Conclusion
Mastering the conversion from micrograms per liter to ppb is less about math and more about confidence. For 99% of water quality scenarios, you can rest easy knowing that 1 μg/L = 1 ppb. It’s a direct translation that makes reading safety reports straightforward.
However, the mark of a true expert is knowing when the rules don’t apply. If you are working with chemicals, oils, or high-salinity water, remember that density is the invisible variable that can distort your data. By keeping the density formula in your back pocket, you ensure that your analysis is not just fast, but scientifically sound.
Next time you review a water quality report, check the units, verify the source, and trust the numbers—you now have the knowledge to interpret them correctly.
❓ Frequently Asked Questions
Is 1 ug/L always equal to 1 ppb?
Only for fresh water and dilute aqueous solutions. Because water’s density is 1 kg/L, the ratio is 1:1. For liquids denser or lighter than water (like oil or concentrated acid), you must divide the ug/L value by the liquid’s density to get the accurate ppb.
How do I convert ppb to micrograms per liter?
It is the reverse process. For water, the value is the same (10 ppb = 10 μg/L). For other liquids, you multiply the ppb value by the density of the liquid (in kg/L) to get the concentration in μg/L.
What is the difference between mg/L and ug/L?
Magnitude. A milligram (mg) is 1,000 times larger than a microgram (μg). Therefore, 1 mg/L is equal to 1,000 μg/L (or 1,000 ppb). Confusing these two is the most common error in water quality reporting.
Why does the EPA use both units?
It often depends on the magnitude of the contaminant. For abundant minerals, mg/L (ppm) is easier to read. For toxic trace elements like lead or mercury, ppb (μg/L) avoids using too many decimal places. They are used to make reports more readable for scientists and the public.
Does this conversion apply to soil testing?
No. Soil is a solid, not a liquid. Soil results are typically reported in μg/kg (micrograms per kilogram). However, conveniently, 1 μg/kg is also equal to 1 ppb on a weight-to-weight basis.



